Introduction
India is the world’s most populated country, and, as such, food security is a huge problem currently the country is confronting. With the development of urbanization and the decrease in available land resources, expanding the utilization of land resources has become an important task to ensure food security. Brinjal or eggplant (Solanum melongena L)., family of Solanaceae, is one of the most significant vegetable crops cultivated worldwide, in tropical and sub-tropical countries. It is a staple in Indian agriculture due to its wide adaptability, productivity, high market demand, and nutritional value, being a rich source of vitamins, minerals, and antioxidants. India is among the leading producers of brinjal, with Chhattisgarh being one of the important cultivation regions, and mostly used as a daily kitchen vegetable for numerous meals. It has been known that it has several therapeutic potentials against disturbances and diseases like gastrointestinal problems, skin-related irritations, dental aches and haemorrhoids. However, despite its commercial and dietary significance, productivity often remains suboptimal due to soil fertility issues, pest incidence, and imbalanced use of chemical inputs.1
The intensive application of chemical fertilizers during the Green Revolution undeniably contributed to enhanced crop yields but when chemical fertilizers exceed the threshold level, they accelerate soil acidification, pollute groundwater, and harm the environment overall. Yet their overuse has led to severe environmental concerns, including soil degradation, groundwater contamination, reduced microbial diversity, and diminished long-term soil productivity.2 This growing concern has shifted global agricultural practices towards more sustainable approaches, such as integrated nutrient management and the utilization of biofertilizers. It is recommended to apply biofertilizers as eco-friendly alternatives as they play a pivotal role in phosphate solubilization, nitrogen fixation, production of ammonia, enzymes, siderophores, and secretion of variable phytohormones.3 Furthermore, they exhibit biocontrol activity against a wide variety of phytopathogenic agents.
Biofertilizers, comprising beneficial microorganisms like Rhizobium, Azotobacter, Azospirillum, phosphate-solubilizing bacteria (PSB) to improve crop performance through multiple mechanisms. Worldwide studies for 20 years on Azospirillum inoculation indicated a 60–70% success with 3–50% improvements in grain yields of crops on account of N fixation and associated root growth stimulation and phyto-hormonesynthesis.4,5 These include nitrogen fixation, phosphate solubilization, siderophore production, and phytohormone synthesis, all of which enhance nutrient uptake and promote vigorous root and shoot development. Unlike chemical fertilizers, biofertilizers actively support soil microbial biodiversity and maintain ecological balance, making them central to the sustainable intensification of agriculture.6
Several studies highlight the advantages of biofertilizers in brinjal production. For instance, integrated use of microbial inoculants with organic amendments has been shown to improve plant height, leaf chlorophyll content, and fruit yield, while reducing disease incidence.7 Moreover, combinations of different biofertilizer strains often exhibit synergistic effects in the rhizosphere, resulting in higher efficiency compared to single-strain inoculations.8 Such synergistic interactions enhance nutrient availability and root colonization, ultimately leading to improved crop growth and yield performance.
The rhizosphere, defined as the narrow soil region directly influenced by root secretions and associated microorganisms, plays a pivotal role in plant–microbe interactions. Soil type and quality strongly influence biofertilizer performance, with rhizospheric soils often exhibiting higher microbial activity than garden soils. Regional variability in soil fertility, such as that found in Raipur, Chhattisgarh, can therefore alter the effectiveness of biofertilizer applications. Since Chhattisgarh’s soils are predominantly characterized by low organic matter and nutrient limitations under rainfed conditions, there is a strong need to develop localized biofertilizer-based strategies to improve brinjal productivity sustainably.9 Effect of Azotobacter, Arbuscular Mycorrhiza on the Growth and Yield of Brinjal. Despite growing evidence supporting biofertilizer use, limited studies have specifically examined the comparative effect of biofertilizer combinations on S. melongena under contrasting soil conditions, such as garden soil versus rhizospheric soil, in the agroecological setting of Raipur. Addressing this knowledge gap is essential, as understanding how microbial inoculants interact with different soil types can help optimize nutrient management strategies and ensure higher yield stability.10
Therefore, the present study investigates the impact of different biofertilizer combinations on the growth and yield of S. melongena L. using garden soil and rhizospheric soil of Raipur, Chhattisgarh. By analyzing growth parameters, yield attributes, and soil health improvements, this research aims to provide insights into sustainable soil fertility management, reduce reliance on synthetic inputs, and contribute to environmentally friendly brinjal production in the region.
Materials and Methods
Collection of Soil Samples
The soils were sampled from a 5–20 cm soil layer and each of them were placed into a sterilized polythene bag and then stored in a refrigerator at 40C in two distinct locations within Raipur district, Chhattisgarh, representing contrasting soil environments to assess the influence of biofertilizer combinations on Solanum melongena L. growth and yield.11 Garden Soil (S1) – The first type of soil was obtained from a managed garden area. Rhizospheric Soil (S2) – The second type of soil, designated as S2, was collected from the rhizospheric zone of actively growing plants. Both soil samples were collected from the topsoil layer (0–15 cm depth), as this horizon is most active in terms of nutrient cycling and root interactions.
Preparation of soil samples
Samples were air-dried, homogenized, and sieved (2 mm mesh) before use in the experimental setup to ensure uniformity. Care was taken to avoid contamination during collection, storage, and handling.
Bacterial Inoculum Preparation
Azospirillum brasilense (B1), Bacillus megaterium (B2), and Bacillus circulans (B3) inoculum were prepared individually and in mixtures.One gram of soil from the root portion was appropriately cleaned out and added to 9 mL of sterile physiological saline Himedia (pH 7.2), vortexed for 10 minutes, and left to rest for 20 minutes. A 100 μL aliquot of each successively diluted soil suspension was then distributed onto the surface of nutrient agar plates and cultured for 2 to 3 days at 30 ± 2°C. To obtain pure colonies, distinct colonies were chosen, subculture on a nutrient agar plate, Himedia, and then preserved in 20% glycerol solutions at -20°C.12
Evaluation for phosphate solubilisation
The positive phosphate solubilising bacterial colonies which was isolated in the media were evaluated. The screening method was carried out to compare the solubilisation efficiency with the other colonies. Finally, the potential colony with high solubilisation efficiency was selected for further characterization.13
Seed Treatment and Pot Experiment
Healthy and uniform seeds were selected and subjected to different biofertilizer treatments prior to sowing. The biofertilizer inoculants used in the study included B1, B2, B3 (beneficial bacterial strains), phosphate-solubilizing bacteria (PSB), and vesicular–arbuscular mycorrhiza (VAM). Individual inoculants as well as their various combinations were prepared to evaluate their synergistic effects on plant growth and soil health. For seed treatment, the required quantity of each biofertilizer was mixed with a suitable adhesive solution to ensure uniform coating. The seeds were thoroughly coated with the inoculum mixture, shade-dried for a specified period to facilitate adhesion and sown immediately after treatment to maintain microbial viability.14 Pot culture experiments were conducted under controlled environmental conditions using two types of soil: garden soil and rhizospheric soil. The soils were collected, air-dried, sieved (2 mm mesh), and homogenized before filling into sterilized pots of uniform size. Treated seeds were sown in the respective pots, and recommended agronomic practices were followed throughout the experimental period.15 Irrigation was provided uniformly to maintain optimum moisture levels. Observations on germination percentage, seedling vigor, plant height, root length, biomass accumulation, and other relevant growth parameters were recorded at predetermined intervals. Soil samples were also analyzed to assess changes in physicochemical and biological properties following biofertilizer application.
Fertilizer application
For the nutrient applications, a combined nitrogen and potassium stock solution was prepared by dissolving 80 g of urea and 80 g of muriate of potash in 400 mL of tap water with continuous stirring to ensure complete dissolution. From this stock, 20 mL was applied uniformly to each designated pot. Phosphorus was supplied from two different sources: triple super phosphate (TSP) and rock phosphate (RP). The TSP solution was prepared by dissolving 20 g of TSP in 200 mL of tap water, and 20 mL of this solution was applied per pot where required. Similarly, 20 g of finely powdered rock phosphate was suspended in 200 mL of tap water, and 20 mL of the well-mixed suspension was applied per pot to ensure uniform distribution. All nutrient applications were carried out carefully and uniformly to maintain consistent distribution among the respective experimental units.16
Results
Physio-Chemical Analysis
Table 1: Different physical parameters of Garden soil and Rhizospheric soil
| SN | Parameters | Garden Soil | Rhizospheric Soil |
| 1 | Texture | Sandy Loam | Loamy |
| 2 | Colour | Reddish Brown | Dark Brown |
| 3 | Moisture Content | Moderate | High |
| 4 | Bulk Density | 1.4 g/cm3 | 1.2 g/cm3 |
| 5 | Porosity | Medium | High |
Garden soil (S1) was found to be sandy loam in texture, whereas rhizospheric soil (S2) exhibited a loamy texture. The garden soil exhibited a reddish-brown color, often associated with iron oxides and moderate organic matter content. On the other hand, the rhizospheric soil appeared dark brown, a typical indicator of higher organic matter and microbial biomass. Since organic matter strongly influences nutrient availability and microbial activity, the darker rhizospheric soil is expected to be more fertile and biologically active, supporting the proliferation of biofertilizer organisms. Moderate moisture content was recorded in garden soil, whereas rhizospheric soil showed high moisture levels. The elevated moisture in rhizospheric soil is attributable to root exudates and greater aggregation, which enhances water retention. Bulk density, which reflects soil compaction and aeration, was higher in garden soil (1.4 g/cm³) than in rhizospheric soil (1.2 g/cm³).17 High bulk density can restrict root penetration, reduce aeration, and hinder microbial activity. Garden soil exhibited medium porosity, while rhizospheric soil showed high porosity. Increased porosity in rhizospheric soil promotes improved water infiltration, aeration, and microbial colonization. The comparative analysis reveals that rhizospheric soil (S2) possesses more favorable physical conditions—loamy texture, darker color (higher organic matter), lower bulk density, and higher porosity—than garden soil (S1). These characteristics are expected to promote higher microbial diversity and activity, thereby potentially enhancing the performance of biofertilizer treatments. By contrast, garden soil represents a more managed but less biologically enriched environment, offering a contrasting baseline for evaluating the effect of biofertilizer combinations.
Table 2: Different chemical parameters of Garden soil and Rhizospheric soil
| SN | Parameters | Garden Soil | Rhizospheric Soil |
| 1 | pH | 6.2 | 5.8 |
| 2 | EC (dS/m) | 0.45 | 0.60 |
| 3 | Organic Carbon (%) | 3.5 | 4.2 |
| 4 | Total Nitrogen (%) | 0.14 | 0.18 |
| 5 | Total Phosphorus (%) | 0.08 | 0.10 |
| 6 | Available Phosphorus (mg/kg) | 20 | 25 |
| 7 | Calcium Carbonate (%) | 12 | 10 |
The garden soil (pH 6.2) was slightly acidic, while the rhizospheric soil exhibited a more acidic nature (pH 5.8). Soil pH is a critical parameter affecting nutrient solubility, microbial activity, and biofertilizer efficacy. A pH range of 5.5–6.5 is generally considered favorable for most vegetable crops, including S. melongena L. The slightly lower pH in rhizospheric soil could enhance the solubilization of micronutrients such as Fe, Mn, and Zn, while also favoring the activity of phosphate-solubilizing microorganisms. The EC of garden soil (0.45 dS/m) was slightly lower compared to rhizospheric soil (0.60 dS/m). Both values fall within the non-saline category, indicating no salinity stress for plants. Organic carbon was higher in rhizospheric soil (4.2%) compared to garden soil (3.5%). Organic carbon content is an important indicator of soil fertility, as it improves cation exchange capacity (CEC), water retention, and microbial proliferation. The higher organic carbon in rhizospheric soil suggests greater microbial biomass and decomposition activity, which are beneficial for biofertilizer performance, as microbes require organic matter as an energy source. Total nitrogen content was recorded at 0.14% in garden soil and 0.18% in rhizospheric soil. Nitrogen is essential for vegetative growth, chlorophyll synthesis, and yield formation. The higher nitrogen levels in rhizospheric soil may be attributed to active biological nitrogen fixation and enhanced mineralization processes. Total phosphorus was 0.08% in garden soil and 0.10% in rhizospheric soil, while available phosphorus was 20 mg/kg and 25 mg/kg, respectively. Phosphorus is crucial for root development, energy transfer, and flowering. Calcium carbonate content was higher in garden soil (12%) compared to rhizospheric soil (10%). Excessive calcium carbonate can lead to nutrient imbalances, particularly by reducing the availability of phosphorus, iron, and zinc due to precipitation reactions.Both garden and rhizospheric soils exhibited only trace amounts of carbonate. This is advantageous for crop growth, as excessive carbonate can cause alkalinity, nutrient precipitation, and reduced phosphorus availability. The trace levels here indicate no risk of carbonate-induced soil constraints. The bicarbonate content was slightly higher in rhizospheric soil (4.2 meq/L) compared to garden soil (3.5 meq/L). Elevated bicarbonate concentrations can contribute to pH buffering capacity, helping stabilize soil acidity levels. Chloride levels were higher in rhizospheric soil (20 mg/kg) than in garden soil (15 mg/kg). Chloride, though often associated with salinity, is an essential micronutrient in small amounts, contributing to photosynthesis and osmotic regulation. Both soils remain well below toxic chloride thresholds, suggesting no risk of salinity stress. Rhizospheric soil showed higher sulfate levels (8.5 meq/L) compared to garden soil (7.0 meq/L). Sulfur in the form of sulfate is crucial for amino acid (cysteine, methionine) and protein synthesis, as well as for enzyme activity. Calcium was abundant in both soils, with slightly higher levels in rhizospheric soil (22 meq/L) than in garden soil (18 meq/L). Magnesium levels were marginally higher in rhizospheric soil (5.0 meq/L) compared to garden soil (4.5 meq/L). Sodium concentrations were low in both soils, with slightly higher values in rhizospheric soil (1.5 meq/L) than in garden soil (1.2 meq/L). Potassium was slightly higher in rhizospheric soil (1.0 meq/L) compared to garden soil (0.8 meq/L). Potassium is a macronutrient critical for stomatal regulation, enzyme activation, carbohydrate transport, and overall stress tolerance. The higher K⁺ availability in rhizospheric soil suggests a stronger nutrient supply to support vegetative growth and fruit development in S. melongena. The ionic composition analysis demonstrates that rhizospheric soil (S2) consistently exhibits higher levels of essential anions (HCO₃⁻, SO₄²⁻, Cl⁻) and cations (Ca²⁺, Mg²⁺, K⁺, Na⁺) compared to garden soil (S1). These enhanced ionic concentrations are likely due to the combined effects of root exudates, microbial activity, and higher organic matter content in the rhizospheric zone. Importantly, all values remain within agronomically safe limits, indicating no toxicity risks.
Germination and Seedling Growth
The germination study revealed a clear influence of biofertilizer treatments on early seed performance. The highest germination percentage was observed in T8 (94%), followed by T5 (92%) and T10 (91%), while the lowest was in T2 (76%)andT6 (78%).In terms of seedling growth, T8 recorded the maximum shoot length (8.6 cm) and root length (7.3 cm), leading to the highest seedling vigor index (1,480), followed by T10 (1,420) and T5 (1,398). The enhanced early growth indicates efficient nutrient mobilization and hormonal stimulation by microbial inoculants. Germination and early seedling development were consistently higher in rhizospheric soil (S2) compared to garden soil (S1), highlighting the positive impact of organic matter and microbial activity in promoting early plant establishment.
Table 3: Effect of different combination of biofertilizers on Germination and Seedling Growth of Solanum melongena L. with garden soil
| Treatments | Germination | Shoot Length (cm) |
Root Length (cm) |
Seedling Vigor Index (SVI) |
| T1 | 74% | 6.2 | 5.1 | 835 |
| T2 | 76 % | 6.4 | 5.3 | 855 |
| T3 | 79 % | 6.8 | 5.5 | 910 |
| T4 | 80 % | 6.9 | 5.6 | 940 |
| T5 | 92 % | 8.3 | 7.0 | 1398 |
| T6 | 78 % | 6.5 | 5.4 | 910 |
| T7 | 86 % | 7.4 | 6.5 | 1190 |
| T8 | 94 % | 8.6 | 7.3 | 1480 |
| T9 | 88 % | 7.6 | 6.7 | 1280 |
| T10 | 91% | 8.1 | 6.9 | 1420 |
| T11 | 89 % | 7.8 | 6.8 | 1320 |
|
T12 |
90% | 8.2 | 7.0 | 1380 |
Table 4: Effect of different combinations of biofertilizers on Germination and Seedling Growth of Solanum melongena L. with rhizospheric soil
| Treatments | Germination | Shoot Length (cm) |
Root Length (cm) |
Seedling Vigor Index (SVI) |
| T1 | 77% | 6.4 | 5.1 | 870 |
| T2 | 79% | 6.6 | 5.3 | 895 |
| T3 | 82% | 7.0 | 5.5 | 980 |
| T4 | 83% | 7.1 | 5.6 | 995 |
| T5 | 93% | 8.5 | 7.0 | 1440 |
| T6 | 80% | 6.8 | 5.4 | 950 |
| T7 | 87% | 7.6 | 6.5 | 1240 |
| T8 | 95% | 8.9 | 7.3 | 1545 |
| T9 | 89% | 7.8 | 6.7 | 1320 |
| T10 | 92% | 8.3 | 6.9 | 1460 |
| T11 | 90% | 8.0 | 6.8 | 1370 |
| T12 | 91% | 8.4 | 7.0 | 1420 |
Garden Soil Experiment
The comparative evaluation of twelve biofertilizer treatments on Solanum melongena L. revealed marked variations in both vegetative growth and yield parameters. Plant height was found to be highest in T3 (18.24 cm), closely followed by T12 (17.66 cm) and T9 (17.24 cm), indicating superior growth performance, whereas T5 (10.28 cm) and T6 (10.27 cm) recorded the lowest values, suggesting limited growth promotion. Branching, which directly influences canopy architecture and fruiting potential, also varied considerably: T6 produced the maximum number of primary branches (20.56), while T3 (18.78) and T12 (18.14) recorded the highest secondary branches, highlighting their role in enhancing shoot proliferation. With respect to fruit morphology, the longest fruits were observed under T2 (19.76 cm) and T8 (19.43 cm), while the widest fruits were produced in T9 (19.96 cm) and T12 (19.55 cm), suggesting that specific treatments favor not only elongation but also fruit girth development. Yield-attributing traits showed that T8 recorded the maximum number of fruits per plant (20.23), followed by T9 (19.09) and T11 (17.53), while T6 and T5 produced the least (<12 fruits per plant). In terms of overall productivity, T11 emerged as the best performer with the highest fruit yield (17.88 g), followed closely by T8 (17.57 g) and T12 (17.56 g), whereas T6 (13.0 g) and T7 (10.26 g) lagged behind despite moderate growth. These findings collectively demonstrate that biofertilizer combinations have a pronounced effect on both vegetative and reproductive parameters of brinjal, with treatments such as T11, T8, and T12 showing superior performance, thereby underscoring the synergistic role of beneficial microbes in enhancing plant growth, fruit set, and yield potential.
Table 5: Effect of different combination of biofertilizers and chemical fertilizer against test plant with garden soil
| Treatments | Plant Height | No. of Primary Branches | No. of Secondary Branches | Fruit Length | Fruit Width | No. of Fruit per Plant | Fruit Yield |
| T1 | 13.4±0.94 | 17.8±0.87 | 9.08±1.26 | 13.08±11.16 | 10.61±0.98 | 15.25±0.66 | 16.5±0.9 |
| T2 | 13.260.61 | 9.61±0.97 | 15.17±1.41 | 19.76±1.11 | 12.06±1.53 | 12.53±1.03 | 17.12±1.02 |
| T3 | 18.24±1.51 | 14.42±1.04 | 18.78±1.16 | 17.16±10.92 | 13.98±0.82 | 15.87±0.73 | 13.93±0.89 |
| T4 | 12.16±0.78 | 18.86±1 | 10.31±1.23 | 16.38±1.8 | 13.35±0.43 | 15.48±1.37 | 13.58±2.4 |
| T5 | 10.28±0.62 | 15.88±0.94 | 13.7±1.46 | 15.25±1.47 | 12.13±0.98 | 11.63±0.89 | 16.96±0.61 |
| T6 | 10.27±1.26 | 20.56±1.66 | 19.53±0.57 | 14.09±11.28 | 11.37±1.12 | 10.06±1.32 | 13±1.39 |
| T7 | 14.9±0.49 | 15.64±1 | 13.88±0.88 | 17.02±1.78 | 11.99±1.12 | 12.01±0.6 | 10.26±0.71 |
| T8 | 14.64±0.84 | 12.65±0.39 | 11.42±1.69 | 19.43±1.29 | 14.54±0.58 | 20.23±0.82 | 17.57±0.56 |
| T9 | 17.24±1.01 | 10.09±2.52 | 12.05±0.56 | 14.98±10.83 | 19.96±0.59 | 19.09±1.73 | 16.03±0.73 |
| T10 | 12.09±1.49 | 17.22±0.52 | 15.88±1.87 | 14.64±1.34 | 13.11±0.59 | 15.81±1.05 | 16.73±0.97 |
| T11 | 16.92±0.5 | 16.07±1.63 | 15.38±0.36 | 18.75±0.52 | 10.58±0.96 | 17.53±0.73 | 17.88±0.92 |
| T12 | 17.66±0.51 | 15.08±1.37 | 18.14±1.04 | 13.43±0.74 | 19.55±0.7 | 15.63±1.73 | 17.56±1.22 |
Data are multiple of three observations
Values ± SD
No. of parameters against treatment are seven
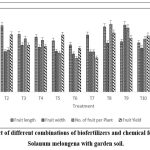 |
Figure 1: Effect of different combinations of biofertilizers and chemical fertilizer against Solanum melongena with garden soil.Click here to view Figure |
Table 6: Effect of different combination of biofertilizers and chemical fertilizers against test plants with rhizosperic soil of agriculture land
| Treatments | Plant Height (cm) |
No. of Primary Branches | No. of Secondary Branches | Fruit Length(cm) | Fruit Width(cm) | No. of Fruit per Plant | Fruit Yield (No.) |
| T1 | 56.24±1.02 | 73.52±1.03 | 66.96±1.31 | 62.96±1.25 | 49.68±1.42 | 49.68±1.13 | 46.74±0.88 |
| T2 | 70.99±1.3 | 63.03±1.33 | 66.24±1.19 | 45.62±1.34 | 74.1±1.15 | 69.97±1.17 | 51.37±1.1 |
| T3 | 50.45±0.82 | 50.5±0.88 | 54.13±0.82 | 60.74±1.25 | 57.96±1.02 | 53.74±1.16 | 63.36±1.44 |
| T4 | 49.18±0.97 | 53.76±1.09 | 55.99±1.33 | 58.68±0.96 | 68.56±0.85 | 50.99±1 | 60.43±0.91 |
| T5 | 62.77±1.45 | 46.39±1.37 | 63.23±1.24 | 50.12±1.41 | 46.95±1.36 | 73.47±0.93 | 73.97±1.42 |
| T6 | 69.25±1.18 | 54.14±1.37 | 47.93±1.43 | 65.53±1.02 | 58.2±0.88 | 48.66±0.96 | 59.86±1.1 |
| T7 | 46.03±1.37 | 72.28±1.4 | 52.76±0.8 | 64.88±1.16 | 54.35±1.09 | 60.6±0.96 | 61.4±0.88 |
| T8 | 50.55±1.04 | 74.09±1.46 | 68.251.03 | 73.18±1.16 | 71.84±1.29 | 62.94±1.05 | 72.66±148 |
| T9 | 47.65±1.47 | 50.88±0.98 | 46.36±1.15 | 54.76±1.01 | 56.66±1 | 53.14±0.83 | 69.86±1.23 |
| T10 | 55.7±1.15 | 53.43±0.84 | 61.28±1 | 49.23±1.44 | 69.07±0.97 | 47.24±0.9 | 74.61±1.14 |
| T11 | 68.17±1.49 | 50.96±0.97 | 45.17±1.27 | 69.46±1.33 | 66.21±0.97 | 66.87±1.31 | 68.14±1.06 |
| T12 | 47.22±1.24 | 55.75±1.24 | 48.48±1.18 | 70.89±0.86 | 63.7±1.38 | 54.93±1.02 | 46.91±0.93 |
Data are multiple of three observations
Values ± SD
No. of parameters against treatment are seven
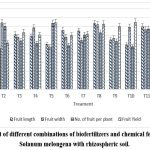 |
Figure 2: Effect of different combinations of biofertilizers and chemical fertilizer against Solanum melongena with rhizospheric soil.Click here to view Figure |
The evaluation of different biofertilizer treatments demonstrated substantial variation in vegetative and reproductive performance of Solanum melongena L., underscoring the influence of microbial inoculation on crop productivity. Plant height ranged from 46.03 cm (T7) to 70.99 cm (T2), with T2 and T6 (69.25 cm) emerging as the tallest, suggesting better nutrient uptake and vigor under these treatments. Interestingly, primary branch numbers were highest in T8 (74.09) and T1 (73.52), indicating enhanced shoot proliferation, while secondary branching peaked in T8 (68.25) and T1 (66.96), highlighting the capacity of these treatments to improve canopy structure. Fruit traits also reflected treatment-specific effects: the longest fruits were produced in T12 (70.89 cm) and T11 (69.46 cm), while the widest fruits were observed in T2 (74.1 cm) and T8 (71.84 cm), signifying that distinct biofertilizer combinations influence fruit morphology differently. In terms of reproductive yield, the number of fruits per plant was maximized in T5 (73.47) and T8 (62.94), translating into superior yields in T10 (74.61 g), T5 (73.97 g), and T8 (72.66 g), whereas comparatively lower yields were recorded in T1 (46.74 g) and T12 (46.91 g). Overall, the treatments T5, T8, and T10 consistently outperformed others by combining optimal vegetative growth, branching, fruit set, and yield potential, thereby illustrating that the synergistic interaction of biofertilizers with soil conditions not only stimulates plant vigor but also translates into higher productivity.
Rhizospheric Soil Experiment
Enhanced growth was noted with T2 (70.99±1.3 cm) in plant height and T8 (74.09±1.46) in primary branches. The highest fruit length (70.89±0.86 cm) and width (71.84±1.29 cm) were observed with multiple inoculations. Fruit yield was maximum with T10 (74.61±1.14 g), highlighting the synergy between biofertilizers and VAM.
Discussion
Solanum melongena L. is an important economic species that exhibits a sensitive response to moderate salinity levels. Several studies have shown an inhibition in growth parameters and biomass production due to the increase in salinity reported that the primary response to salinity stress is expressed by a reduction of root and shoot development.18
The present study demonstrated that different biofertilizer treatments significantly influenced the growth and yield of Solanum melongena L. across the measured parameters. Treatments such as T5, T8, and T10 consistently outperformed others, reflecting higher plant height, improved branching, larger fruit size, and greater yield. These findings align with the well-established role of biofertilizers in enhancing nutrient availability and stimulating plant growth through microbial-mediated mechanisms. Biofertilizers such as nitrogen-fixing bacteria, phosphate-solubilizing microorganisms, and potassium-mobilizing bacteria improve soil fertility by converting unavailable nutrients into plant-accessible forms, thereby enhancing vegetative and reproductive performance.19,20
The observed improvement in plant height and branching under T8 and T5 treatments can be attributed to better root development and hormonal stimulation by microbial inoculants, particularly auxins and cytokinin produced by plant growth-promoting rhizobacteria (PGPR). Similar findings were reported by who demonstrated that integrated biofertilizer application in brinjal significantly enhanced plant height and branch proliferation.21 The higher fruit length and width observed in treatments T2, T11, and T12 suggest that nutrient availability during reproductive stages played a crucial role in determining fruit morphology, corroborating the work of, who noted that balanced microbial activity improved assimilate partitioning towards fruit development in Brinjal.22
In terms of yield performance, the superior outcomes under T5, T8, and T10 can be linked to the synergistic action of multiple microbial consortia. Previous studies have highlighted that the combined use of Azotobacter, phosphate-solubilizing bacteria, and vesicular-arbuscular mycorrhiza enhances yield attributes more effectively than single inoculants.23 Phosphorus is a crucial macronutrient that is required by the plants to augment their plant growth. This condition leads to the high usage of chemical fertilizers of P which subsequently increases the cost of chemical fertilizers. Furthermore, the higher fruit number per plant in these treatments underscores the role of improved nutrient cycling and better soil structure, as noted by where biofertilizers were found to reduce bulk density and improve water retention, thereby favoring reproductive success.24
The variation among treatments also emphasizes the importance of soil type and microbial compatibility. Rhizospheric soil, characterized by higher organic carbon and nutrient content in this study, likely provided a more favorable environment for microbial colonization and nutrient transformation compared to garden soil, which may explain the better performance of treatments applied under rhizospheric conditions. This observation resonates with findings by who argued that the efficacy of biofertilizers depends not only on the microbial strains used but also on the soil’s physicochemical properties.25
Conclusion
The present investigation highlights the significant impact of biofertilizer combinations on the growth and yield performance of Solanum melongena L. under the contrasting soil conditions of garden and rhizospheric soils in Raipur, Chhattisgarh. Treatments such as T5, T8, and T10 consistently outperformed others, showing superior results in plant height, branching, fruit morphology, and overall yield, underscoring the efficacy of integrated microbial inoculants. The rhizospheric soil, with its higher organic carbon, nutrient availability, and favorable physicochemical characteristics, supported greater plant performance compared to garden soil, emphasizing the critical role of soil environment in enhancing biofertilizer efficiency.
These findings demonstrate that biofertilizers not only improve nutrient uptake and soil fertility but also act as sustainable alternatives to chemical fertilizers by enhancing crop productivity while maintaining ecological balance. The synergistic effects of multiple microbial consortia were particularly evident in yield improvement, highlighting the importance of integrating different functional groups of biofertilizers for maximum benefit. Thus, adopting biofertilizer-based nutrient management can serve as a sustainable strategy for improving brinjal cultivation in Chhattisgarh and similar agro-climatic zones. Future research should focus on long-term field trials and microbial diversity studies to optimize biofertilizer formulations and their compatibility with local soil systems for consistent and scalable agricultural productivity.
Acknowledgement
The authors are grateful to MATS University, Raipur for providing valuable information.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Author Contributions
Nomesh Kumar Jethmal: Conceptualisation, Methodology, Writing – Original Draft.
Sounak Sarkar: Writing – Writing original draft & Editing.
References
- Kumar A, Singh R, Yadav AN, Bishnoi R, Mishra S. Harnessing plant microbiomes for sustainable agriculture. Front Sustain Food Syst. 2020;4:1-15.
- Swain MR, Arora NK, Ray RC. Biofertilizers in agriculture: recent advances and future outlook. Appl Soil Ecol. 2021;164:103911.
- Amoo AE, Enagbonma BJ, Ayangbenro AS, Babalola OO. Biofertilizer: an eco-friendly approach for sustainable crop production. In: Food Security and Safety: African Perspectives. Cham, Switzerland: Springer International Publishing; 2021:647-669.
CrossRef - Katiyar P, Kumar S, Arora NK. Interactions of nitrogen-fixing bacteria and cereal crops: an important dimension. In: Nitrogen Fixing Bacteria: Sustainable Growth of Non-Legumes. Singapore: Springer Nature Singapore; 2022:169-194.
CrossRef - Goel R, Singh R. Growth promotion and response of staple crops to inoculation with Azospirillum: a review. Curr Trends Biotechnol Pharm. 2025;19.
CrossRef - Parmar P, Sindhu SS, Kumar V. Effect of biofertilizers and organic amendments on plant growth and soil properties. Environ Sustain. 2020;3(1):11-20.
- Elnahal AS, El-Saadony MT, Saad AM, Desoky ESM, El-Tahan AM, Rady MM. The use of microbial inoculants for biological control, plant growth promotion, and sustainable agriculture: a review. Eur J Plant Pathol. 2022;162(4):759-792.
CrossRef - Mishra J, Singh R, Arora NK. Biofertilizers for sustainable agriculture: an overview on recent advances. Biocatal Agric Biotechnol. 2021;26:101718.
- Prakash MWH. Effect of Azotobacter, Arbuscular Mycorrhiza on the Growth and Yield of Brinjal [dissertation]. Rahuri, Maharashtra, India: Mahatma Phule Krishi Vidyapeeth; 2024.
- O’Callaghan M, Ballard RA, Wright D. Soil microbial inoculants for sustainable agriculture: limitations and opportunities. Soil Use Manag. 2022;38(3):1340-1369.
CrossRef - Uma K. Innovative Exploration of Crop Beneficial Bio-Agents for Reduction of Insecticidal Residues in Vegetable Grown Soils of Surguja District of Chhattisgarh [dissertation]. Raipur, Chhattisgarh, India: Indira Gandhi Krishi Vishwavidyalaya; 2020.
- Da Silva Oliveira CE, Fernandes GC, Jalal A, Sant’Ana GR, Aguilar JV, de Camargos LS,. Inoculation and concentration of Bacillus subtilis and Azospirillum brasilense to nitrogen metabolism, leaf gas exchange and plant growth in hydroponic lettuce. Int J Veg Sci. 2025;1-24.
CrossRef - Wang Z, Zhang H, Liu L, Li S, Xie J, Xue X, Jiang Y. Screening of phosphate-solubilizing bacteria and their abilities of phosphorus solubilization and wheat growth promotion. BMC Microbiol. 2022;22(1):296.
CrossRef - Patyal D, Sachdeva K, Sharma K, Khan RR. An innovative and sustainable seed coating technology for improving seed quality and crop performance. J Sci Res Rep. 2025:597-607.
CrossRef - Adamczewska-Sowińska K, Wojciechowski W, Krygier M, Sowiński J. Effect of soil regenerative practice on selected soil physical properties and eggplant (Solanum melongena) yield. Agronomy. 2022;12(7):1686.
CrossRef - Chatzistathis T, Sarropoulou V, Papaioannou E, Giannakoula A. Inorganic and organic fertilization effects on the growth, nutrient uptake, chlorophyll fluorescence and fruit quality in Solanum melongena plants. Agronomy. 2025;15(4):872.
CrossRef - Bodner G, Mentler A, Keiblinger K. Plant roots for sustainable soil structure management in cropping systems. In: The Root Systems in Sustainable Agricultural Intensification. Cham, Switzerland: Springer International Publishing; 2021:45-90.
CrossRef - Castillo JM, Mancilla-Leytón JM, Martins-Noguerol R, Moreira X, Moreno-Pérez AJ, Muñoz-Vallés S, . Interactive effects between salinity and nutrient deficiency on biomass production and bio-active compounds accumulation in the halophyte Crithmum maritimum. Sci Hortic. 2022;301:111136.
CrossRef - Ramesh A, Sharma MP, Sharma SK, Yadav N. Biofertilizers: potential approach for sustainable agriculture. Int J Curr Microbiol Appl Sci. 2019;8(4):1245-1256.
- Chand S, Indu, Singhal RK, Govindasamy P. Agronomical and breeding approaches to improve the nutritional status of forage crops for better livestock productivity. Grass Forage Sci. 2022;77(1):11-32.
CrossRef - Sahu R, Thakur A, Nag S. Effect of integrated nutrient management on growth and yield of brinjal (Solanum melongena). Int J Curr Microbiol Appl Sci. 2018;7(2):1620-1626.
- Singh SK, Pachauri RK, Khatoon H, Katiyar D, Agnihotri G. The role of biofertilizers in enhancing soil and productivity: a review. Int J Plant Soil Sci. 2025;37(3):141-161.
CrossRef - Masrahi AS, Alasmari A, Shahin MG, Qumsani AT, Oraby HF, Awad-Allah MM. Role of arbuscular mycorrhizal fungi and phosphate solubilizing bacteria in improving yield, yield components, and nutrients uptake of barley under salinity soil. Agriculture. 2023;13(3):537.
CrossRef - Imran, Ortas I. Mechanism in soil health improvement with soil microbial actions and its role in potential agriculture. Commun Soil Sci Plant Anal. 2025;56(13):2066-2087.
CrossRef - Singh V, Prasad VM. Role of biofertilizers in sustainable cultivation of brinjal (Solanum melongena). J Pharmacogn Phytochem. 2021;10(1):1590-1594.


0 Replies to “Assessing the Synergistic Effect of Biofertilizers and Phosphate-Solubilising Bacteria on Brinjal Solanum melongena L. in Chhattisgarh’s Garden and Rhizospheric Soil”