Introduction
Microbial populations are crux of the soil environment that interfaces plants in a number of ways, the most significant of which is through the root area. Soil degradation has been witnessed due to excessive use of chemical fertilisers, for which organic materials should be used as a supplement to chemical fertilizers to enhance maintain soil productivity.1,2 Solid, liquid and granular biological based biofertilizer is a low expensive and environmental friendly apart from supporting soil health, plant growth and productivity. Biofertilizers are defined as “products containing carrier based (solid or liquid) living microorganisms which are agriculturally useful in terms of nitrogen fixation, phosphorus solubilization, or nutrient mobilization”.3 Nitrogen-fixators, phosphate- and potassium-soluble fertilizers, phosphorus-mobilizing fertilizers, biofertilizers for secondary macro and micronutrients, zinc- and iron-soluble fertilizers, plant-growth-promoting rhizobacteria (PGPR), and compost are the different categories of biofertilizers based on the microorganisms and the advantages they provide.4 Basic requirements of substrate to support the shelf life of bacteria and fungi viability in long duration maintain the microbial population with ensure from lab to field rhizosphere profile for healthy of soil profile and suitable to plant growth.
Main Motto on Carrier Material
Maintain suitable moisture retention and comparably growth pattern of microbial communities.5
An adequate maturity and prevent the unfavourable stress to carrier material with soil physico-chemical properties stability.6
The carrier of the inoculant refers to a solid, semi-solid or liquid substance in which certain bacteria or microorganisms can maintain a certain amount for a period of time.7 According to Shilpa and Brahmaprakash, a carrier material’s ability to support a larger population of injected microorganisms during extended storage times is one of its most important characteristics. Inoculants for bacteria, fungus, algae, and actinomycetes are made using a range of carriers. Among them, common organic transporters include sawdust, lignite, charcoal, peat, pressed mud, loam, and cow dung cake. Carriers help microbial inoculants survive and develop by transporting live inoculants from the lab to the rhizosphere. High organic content, water retention, neutral pH, non-toxicity, affordability, and ease of processing are characteristics of ideal carriers. Compost or vermicompost improves field performance under environmental stressors, seed adhesion, and storage stability.8 The biological-based fertilizer works by including beneficial living organisms that enhance plant health and biomass while also providing strong support for soil fertility and environmental friendliness.9
Role of Carrier on Performance of Microbial Culture
In recent years, great efforts have been put forth to develop environmentally friendly formulations of pesticide products. Vast variety of materials that can be added to improve the effectiveness of pesticides, such as clay minerals, silicate materials, polymers, and research that extends from organic and inorganic materials (Figure 1). Biofertilizers are prepared as carrier-based inoculants which enable easy handling, long-term storage and effectiveness. Appropriate carrier materials provide a suitable environment to extend the life of microorganisms. Therefore, in the unique production of biological fertilizers, organisms can be used at the farm level for a long time in multiple cropping sequences. Symbiotic root interactions between nitrogen-fixing rhizobia and carrier-based inoculants improve soil fertility and legume development. Rhizobia transform air nitrogen into a form that may be used by forming nodules on roots. Through enhanced nutrient availability and stress resilience, they also generate phytohormones and enzymes that support plant health, productivity, and sustainable agriculture.10 Solid, granular, capsule and liquid based biofertilizer available marketable throughout world, efficiently support particularly leguminous crop suitable solid based biofertilizer favourable to enhance rhizobia species.11,12 Investigation of Kumar et al., the survival and phosphate-solubilizing efficiency of phosphate solubilizing bacteria (Bacillus and Pseudomonas spp.) using various carrier materials (peat, lignite, charcoal, and vermiculite). They found that peat and lignite supported higher viability and sustained phosphate solubilization activity over six months compared to other carriers.13,14 A carrier should need a carbon sources to produce biofertilizer. One of the important things to choose a material as carrier due to its capability to maintain population of inoculant. Inoculum formulations using carrier material have widely been used to facilitate the introduction of high cell number and increased survival of microorganisms in soil.15 At present, beneficial bacteria are used in a variety of agro-economically significant plants as both liquid and solid carrier-based inoculant preparations. However, the bacterial strain utilized to produce the inoculant has a major impact on the carrier-based formulation’s performance (Figure 1).16 The survival rate of microbial inoculants introduced into the soil profile system on depend physic-chemical parameters such as moisture level, heat and nature of carrier material. The substrate acts as a catalyst for the shifting of micro size organism from the control condition to the rhizosphere zone of agriculture fields.17 Successfully prepared different organic based carrier material formulation with beneficial microbes such as farmyard manure, vermi-cost and microbial compost.18 In the development of microbial inoculants, the incorporation of organic materials as carrier amendments has been shown to enhance the overall quality of the inoculant formulations. These improvements include increased adhesion to seeds, greater product stability, enhanced viability of beneficial bacteria and fungi during storage, and improved adaptation to soil environments under both biotic and abiotic stress conditions. The use of various additives, selected for their physicochemical properties such as acidity, toxicity profile, relaxation time, and high water retention capacity, is common in inoculant production.19, 9
Recent studies of Díaz-Rodríguez et al, have demonstrated that biologically derived composts can serve as effective substrate materials for biofertilizer production.19 However, comprehensive reviews on the role of compost as a substrate-particularly its capacity to supply macro- and micronutrients to the soil while supporting the persistence of microbial populations-remain limited. Experimental approaches have involved the use of vermicompost and urban composts as modifiers for traditional carriers like talc powder, aiming to create formulations that support diverse microbial communities and improve the viability of inoculated cultures. Conventional carrier substrates for biofertilizer production, such as peat, lignite, and talc, are widely recognized for their ability to promote the growth and survival of microbial inoculants. The addition of organic fertilizers, including earthworm compost and urban compost, as carrier components has been found to further extend the viability of beneficial microorganisms in these formulations. This is primarily attributed to the provision of carbon and nitrogen sources, which sustain the metabolic activity of the inoculated microbes during storage and application. Overall, the integration of organic amendments into carrier systems represents a promising strategy to enhance the efficacy and shelf life of microbial inoculants in sustainable agricultural practices.19,13
 |
Figure 1: Organic based carrier material development processesClick here to view Table |
Ingredients of Carrier Material
Numerous carrier materials have been shown to be effective, such as pressed sludge, calcium alginate, charcoal soil mixture, wheat bran,20 peats, and soil implant formulations based on phosphate rock suggested by Viveganandan and Jauhri,21 And Viveganandan and Jauhri,22 Research on inexpensive and readily accessible waste materials as carriers for biological vaccines is currently ongoing since phosphobacteria and nitrogen-fixing bacteria have a long shelf life.14 Biofertilizer formulation mainly depend upon suitable substrate material (Carrier) to multiplication rate of plant growth promoting microorganism, because during plant growth require nutrients, free of biotic stress, abiotic stress, soil water holding capacity, soil infiltration rate they can augment the shelf life of PGPR strains. A suitable carrier should be non-toxic, sterility, non-reactive, higher proposition nutrient ratio, humidity with sterile charcoal, lignite, peat, farm yard manure (FYM), and rice husk have been tested as carriers.23 Alginate beads, wet and dry alginate microbeads, and gum-arabic preparations of bacterial species like Azospirillum brasilense Cd, Pseudomonas fluorescens, and Rhizobium sp. have been evaluated, as have the drawbacks of direct inoculation and the use of different solid-phase bacterial inoculants.24-26 Over a six-month period, the vitality of bacterial inoculants in all formulations was assessed while they were stored at 40°C and room temperature. Compost has been proposed by a number of scientists as a biofertilizer carrying medium. However, very little study has been done on the function of high-quality compost in preserving the microbial community.27 Numerous studies have focused on enhancing the survival of microorganisms in carrier materials by incorporating various amendments. The addition of different substances such as soya meal and soybean powder, molasses, farmyard manure, composted straw powder, teak leaf powder, and mannitol sucrose molasses, which is rich in nutrients and serves as a readily available carbon source, has been shown to support the viability of beneficial bacteria like Rhizobium spp during storage.28,8
 |
Figure 2: Strategies for the type of Carrier materialsClick here to view Figure |
Inorganic Carrier
Inorganic carrier based substrate such as zeolite, CaCO3 powder, lime, clay, silica, pearlite, alginate and lignite (Figure 2) with beneficial to the growth of microbial inoculants, but problem reduce moisture content in solid based substrate material like such as zeolite and CaCO3 powder as zeolites are natural hydrated aluminium silicates, which can be used agroforestry systems due to their viability to retain soil moisture and minimize nutrient loss.29
Microbial Shelf-life in Zeolite Carrier Material
The moisture of the organic matter is 22.3%, and the latter absorb moisture and then decreases when the zeolite is added. The solid formulations used by zeolites ensure cell protection by the adsorption of the zeolite surface. The volume and surface area of the fine pores of the zeolite powder are greater than 208 m 2 g of surface of the clay soil around 0.25 cm 3 / g and 754 m 2 / g reported. Bacterial cells are adsorbed on the surface of the zeolite and protect them from the harsh environment. The great adherence of bacteria to zeolites was less than glut rapid bacteria because the structure of the cell wall was different.30 The composition of solid inoculation affected the population of Azotobacter on 7th and 14th days. On 7th day, the addition of 5% zeolite resulted in an increase in the Azotobacter population, but on 14th day, the increase in their number depended on the extent of the initial increase. Similar to the population of Bacillus, the ingredients of zeolite and liquid inoculators in solid bio-fertilizers did not affect the populations of Azotobacter during the period of 21 and 28. In observation highest population of Azotobacter in the 28th-day-old substrate was observed in the treatment using five solid bio-fertilizers initially inoculated with liquid inoculum concentrated in 5% zeolite, followed by treatments with 1% zeolite and 10% liquid inoculum. To maintain the viability of these solid-inoculated cultures, it is essential to store them under consistent room conditions for at least six months. Although the bacterial population typically declines over this period, studies have shown that the population of a single-strain inoculator can actually increase after three months of storage.31 On the contrary, after 3 months after storage, the decrease in the bacterial population of solubilized bacteria of the slaughtered root mucosa and phosphate was significant after storage of 3 months.32 In general, vaccination concentrations with zeolite and the first liquids were not statistically important, but increased bacillus of 7 days and the number of Azotobacters. Two different solid based biofertilizer composition: 1% zeolite and a liquid inoculum at 10% and at 5% zeolite and a 5% liquid inoculum. Both formulations retain the logarithm of CFU / g, respectively, 12 and 10 pairs of CFU / g, respectively, more than the bacterial population of solid bio-fertilizer Indonesia.
Talc Carrier Materials
Talc based solid biofertilizer formulation to store up to 180 days for self life viability, plant root colonize ability and plant growth promotion ability under normal and stress condition. Previous data demonstrated that both carrier’s peat and talc provided comparable results for Pseudomonas strain at the majority of time periods and had a shelf-life of up to 180 days. Cell viability in peat for 180 days was comparable for LBUM677 and LBUM223 (3 × 107 and 5 × 107 CFU g-1, respectively), while LBUM677 with talc produced superior results than LBUM223 (2 × 108 and 4 × 107 CFU g-1, respectively).33 Compared to talc-based formula and clay-based formula(80 and 9.3 x 108 CFU, respectively), all cellulose-based formulas with and without enrichment can keep the viable cell count higher and the average cell count over the entire storage period. 81.7 x 108 CFU / g formula / g, respectively, after 180 days of storage. The log CFU g-1 of talc, almond and almond flour of populations of Enterococcus faecalis was 8.0 to 8.5, 4.8 to 5.8 and 4.9 to 5.4 log CFU g-1 , respectively. DI almonds (100 g batch) retained 1.4 ± 0.2 g of inoculated talc, or approximately 1% of the talc used in the inoculation, these inoculated almonds produced approximately 6 log CFU g-1 of Enterococcus faecalis. Deionized almonds contain 4.8 to 5.8 log CFU g-1 of Enterococcus faecalis, which is expected to be less because some talc will remain on the sieve. Best efficient viability of talc-based substrate highly supports at room temperature, Pseudomonas strains can persist for several months, remain to colonize the rhizosphere, and carry out advantageous plant-beneficial activities.29
Organic Carrier Materials
Organic carrier materials such as biochar, wood ash (soft wood and hard wood), fly ash, Sawdust, Paddy straw powder, Wood charcoal, peat, rice grain, wheat bran, farmyard manure (FYM), poultry manure, soya meal & soybean powder, molasses, biogas sludge, Spent mushroom substrate (SMS), vermiculite, composted straw powder, teak leaf powder, mannitol and sucrose (Figure 2).
Fly Ash
India produces more fly ash than any other country, but it only uses a tiny percentage of it, whereas European nations use almost all of it in relation to their overall energy use. Thermal power plant waste of fly ash to amendments of poor physical-chemical and microbial parameters soil, to enhance the soil physical parameters such as porosity, infiltration rate and texture. Improvement chemical properties such as pH, EC (Electrical Conductivity), macro, micro elements solubility and increase population of microbes by application of fly ash.34 On previous work fly ash supported a field trial on onion evaluated AM fungi, A. chroococcum, and fly ash (25, 50, 100 t/ha) with NPK. The 25 t/ha fly ash + 50% NPK + biofertilizers showed highest yield (43.3% over control), improved growth, phosphorus availability, and mycorrhizal dependency, proving cost-effective and eco-friendly for nutrient management.
Shelf-life and Performance of Microbes with Fly Ash Carrier
Different macro and micro components, as well as a zero microbial load, are present in the unsterilized fly ash substrate. According to research studies, varying fly ash concentrations (10–100%) can significantly increase the fungal population up to the 45th day of incubation without affecting the native microbial population. Similarly, a 1:1 ratio of soil and fly ash is appropriate for microbial life, including A. brasilense, A. chroococcum, and B. circulans, and fly ash alone demonstrated its maximum viability in fly ash alone, with a population increase of 80–100 times on the 75th day, followed by soil: fly ash.35 As reported in a previous study35 Juwarkar and Jambhulkar, similar to our findings, the application of fly ash in a 1:1 ratio enhances the growth of P. striata in soil by 100-fold. By the 75th day after planting, the population becomes well established. The addition of organic material to fly ash complexes the heavy metals, reducing their toxicity.36 Azotobacter spp. positive produces siderophores that binds iron and molybdenum controlling their concentration in soil presence of organic matter has an additive effect as it reduces the concentration of toxic metals through sorption, lowers the C/N ratio and provides organic compounds, which promote microbial proliferation and diversity.37 According to Parab et al.34, fly ash has a beneficial effect on the root infection of both native and injected mycorrhizal fungi. Additionally, they demonstrated that the mycorrhizal dependence of onions fluctuates with fly ash additions, peaking at a fly ash dosage of 25 mg ha-1, which also coincided with the best crop output. Long-term use of fly ash38 in conjunction with organic fertilizer such as compost and Azospirillum greatly boosted the amount of P, K, Ca, Mg, S, and Si that was accessible, according to Sevalkumari et al.. Applying 20 mg ha-1 of fly ash together with FYM at 12.5 mg ha-1 and chemical fertilizers produced a similar result.
Merits of Fly Ash
As a substitute for chemical fertilizers, many macrogenous flying macros and ashes can be used. It has been noted that using flying ash to cultivate Azotobacters can cut the need for chemical fertilizers by as much as 50%. In addition, the best meetings of AM bacteria, phosphorus solubilized microorganisms and actinomycetes will further improve soil sustainability. Indirectly, it will help you contamination of soil, emergency emissions to environments by inorganic fertilizer. Industry, and cost reduction.39
Fly Ash based Carrier for Biofertilizers, Biopesticides and Diseases Control
Azotobacter formulations are liquid-based and come in a variety of carriers. These transport materials, however, raise expenses without offering the soil and plants any further advantages. They also exhaust natural resources. Applying fly ash as an Azotobacter carrier can be a practical method of managing it while also lowering production material costs and environmental impact.34 According to Gaind and Gaur, the best options for diazotroph carriers were fly ash by itself or a soil: fly ash blend (1: 1).35 It has been demonstrated that up to 50% fly ash, soil, and other organic fertilizers, particularly vermicompost are effective in enhancing Azotobacter bacterial cell density and shelf life.40
Fly ash and Azotobacter together can lessen the need for costly chemical fertilizers, which have a negative impact on soil quality. Additionally, fly ash works well as a dust insecticide and a suitable carrier for herbal pesticides.41 Sankari and Narayanasamy, assessed the bio-efficacy of fly ash as a carrier for pests of rice, brinjal, and okra, including Azadirachta indica, Eucalyptus globulus, Vitex negundo, Curcuma domestica, Capsicum annum, Piper nigrum, Acorus indica, and Ocimum sanctum.42 Vermicompost and fly ash work well together to control pests and weeds on a variety of vegetables.43 Fly ash is rich in silica which is beneficial for plant growth, overcomes abiotic and biotic stresses by preventing shedding (falling) and Azotobacter has antifungal and antibacterial properties that increase resistance of plants to pests, diseases, as well as other stresses. According to an assortment of studies, applying 50 mg ha-1 of fly ash considerably boosts the production of a number of crops, including red gram, mustard, rice, wheat, potatoes, and maize.44 When Azotobacter is inoculated with 50% chemical fertilizer on onions in rabi crops at 50 Mg ha-1 fly ash, there might be a 40% rise.45 40% fly ash was shown to have a nematoidal effect and was recommended for nutrient provision and the treatment of tomato root-knot disease brought on by Meloidogyne sp.46,47 Cultivars of tomatoes cultivated in soil treated with fly ash were more resistant to the wilt fungus Fusarium oxysporum.47 The use of fly ash was found to inhibit the growth of seven soil-borne pathogenic bacteria by Karpagavalli and Ramabadran,. When used in conjunction with organic and biofertilizers, fly ash amendments in agriculture offer several advantages, including improved crop quality, soil fertility, insect and disease management, and crop yield. Bacillus thuringiensis, or Bt, strains and subspecies are the most often employed microbial insecticides. This bacterium kills one or a few related species of insect larvae, and each strain generates a unique combination of proteins. Some Bt’s are particular to flies and mosquitoes, whereas others control moth larvae that are present on plants. Biochemical pesticides are naturally occurring compounds that use non-toxic methods to manage pests. In contrast, traditional insecticides are often made of synthetic ingredients that either directly kill or inactivate the bug.48
Fly Ash with Vermicompost Carrier Material
By adding organic, chemical, and/or biofertilizers, fly ash’s effectiveness can be increased. Vermicomposting fly ash has also been proven in studies to increase the availability of several nutrients.49 Vermicomposting is a technique that uses the microorganisms found in earthworms’ guts to break down organic materials quickly. It has been shown to improve nutrient availability and microbial activity in soil. According to a number of studies, earthworms such as Lampito mauritii, Eudrilus eugeniae, and Eisenia foetida may be able to turn fly ash into the best manure when combined with different amounts of cow dung.49-52 The ideal ratio for vermicomposting was found to be up to 50% fly ash with cow manure.53
Vermicomposting
NPK and micronutrients are abundant in the vermicompost.54 Additionally helpful alternatives to inorganic fertilizers that enhance soil quality include vermicompost and biofertilizer. These bacteria can fix atmospheric nitrogen or solubilize insoluble phosphorus and make it accessible for crops. Biofertilizers are live organisms that can mobilize nutrients from unusable form through biological processes. Azospirillum, Phosphobacteria, VAM, Azotobacter, Rhizobium etc., are the main types of organisms widely recommended for many field crops. According to Ashwin et al., vermicompost helped fungal inoculants grow, supporting populations of 1.2 x 107/g and 0.009 x 107/g for up to 420 days in A. awamori at 20% and 25% moisture levels, respectively. Up until 90 days, the population of the zinc-solubilizing fungus A. niger increased consistently; after that, it gradually declined. In this case, a 25% moisture level sustained the A. niger population over time more effectively than a 20% moisture level.55
Merits of Vermicompost
Vermicompost produced by earthworms, is nutrient-rich, retains moisture well, and contains beneficial enzymes and plant hormones. Its high microbial activity and ability to enhance nutrient availability make it an excellent carrier for biological inoculants, supporting their survival, growth, and function, thus promoting sustainable crop productivity and soil health.56 As a handy support material, vermicompost can help fungal inoculants (such Aspergillus awamori and Aspergillus niger) last up to a year. Applying earthworms, which are abundant in beneficial microorganisms, in the field is a simple way for farmers who practice sustainable agriculture to increase crop yields.55
Biochar
Bacteroidetes which are engaged in the organic matter cycle, tended to decline as a result of the carrier-based inoculation. Therefore, it may be assumed that fixing Bacillus sp. on charcoal prior to inoculation improved soil carbon and nutrient availability and decreased metal (loid) stress. In fact, its aeration qualities, high porosity, and strong water-holding capacity define it. Carbon sequestration and soil fertility have been enhanced by the use of biochar as a soil supplement. Because biochar has a high carbon content, strong nutrient value, and no harmful compounds, it can serve as the ideal substrate for helpful microbes, particularly heterotrophs due to the numerous auxiliary advantages and effective potential of using biochar to manufacture bioinoculants in India.57 When the carrier was inoculated with Azospirillum broth, the highest moisture content of acacia charcoal produced in the UK after 180 days of storage was 25.33%, which was significantly higher than the 24.32% of the brown coal-based Azospirillum inoculant. The study revealed that the use of acacia charcoal from the United Kingdom can prolong the survival time of microbial inoculants for up to six months.
Carrier based Biocontrol Agent
Bacterial formulation is a product that consists of bacterial cells and a kind of preservative which use to control soil borne diseases. The bacterial component of formulation needs to survive for a considerable length of time and to have in vivo efficacy.58 The shelf-life of environmentally sensitive micro-organisms continues to be a challenging and successlimiting step in development of biocontrol products.59 While Pseudomonas fluorescens has significant potential as biocontrol agent, it is very sensitive to environmental factors, in particular temperature and acidity.60
Self-life of Biochar
Research indicates two distinct aspects: the formation of the consortium and the preparation of the carriers. The carrier substance is the primary determinant of any microbial inoculant’s survival. The research’s carrier material satisfies the fundamental requirements for an excellent carrier, including neutral pH, high organic content, water-holding capability, ease of availability, and affordability.61 The biggest viable cells of the Japanese Brachyrhizobium, Pseudomonas fluorescens, and Bacillus megaterium in the single, double and triple inoculants do not alter when compared to the talcum powder formula in the worm manure modified talcum powder and urban compost formula. This could be because organic materials contain a lot of nutrients that help microbial inoculants survive while being stored in rhizospheres.17 Similar findings were found by Rajasekar and Karmegam, (2010), who stated that over 1 X 107 viable cells/g of Chromococcus, Azotobacter, and Bacillus megaterium62 were seen in various combinations of earthworms and lignite for a maximum of 10 months. Pseudomonas fluorescens, Bacillus subtilis, and Azospirillum brasiliensis have been shown to have a high survival rate in a variety of organic carriers, including vermicompost, waste fungus, free-range fertilizer, pressed sludge, and rice bran.63,27,64 The highest survival rate of microbial inoculants among all vector-based formulations is observed in triple-layer carrier systems. These carriers provide microenvironments for microorganisms and serve as media for the development of biological products that meet desired quality standards and offer extended shelf life. In addition to the various types of carriers,65 it has been found that storage temperatures significantly affect the acceptability of agricultural products and the shelf life of bioinoculants.26 Many types of beneficial bacteria have a longer shelf life after being stored at lower temperatures, which has been supported by numerous investigations.66,67
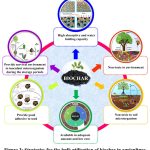 |
Figure 3: Strategies for the bulk utilization of biochar in agricultureClick here to view Figure |
 |
Figure 4: Microbial Culture development and Inoculated in Carrier Materials. Click here to view Figure |
Conclusion
According to the review’s findings, microbial culture transfer applying an organic-based substrate successfully adapts a variety of N, P, and K mobilisation bacteria and actinomyces for usage with carrier materials such fly ash, vermicompost, and biochar. Zeolite and talc powder are the most common inorganic-based carrier materials used to incorporate biofertilizer microorganisms. Better performance and cost-effectiveness with a sustainable level employing a variety of organic substrates that are readily available locally and rapidly and that have a long self-life, are compared to inorganic carrier materials.
Acknowledgement
The authors would like to thank Department of Agriculture, Kalasalingam Academy of Research and Education (KARE), Krishnankoil-626126, India which provided grant support for the literature collection and carry out the research work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
No conflict of interest for each author.
Data Availability Statement
The document used to support the review of this paper is available from the corresponding author upon reasonable request.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Ethics Approval Statement
This study does not involve an experiment on humans and animals.
Authors’ contributions
Kannan Raju – Conceptualization, Methodology, Writing – Original Draft.
Seenivasa Moorthy Thirupathi – Software and statically Analysis, Writing – Review & Editing.
Sheik Abdul Rahuman Mohammed Abdul khader – Data Collection and Analysis
Shruthikaa Pugalenthi, Sanjay Kishore Kumar, Balammal Senthil Vel, Shobicka Sri Malarmaran Sivamalar and Swathi Marichamy – Field evaluation, Crops monitoring and data collection,.
References
- Kanu, S. A., Okonkwo, J. O., Dakora, F. D. Aspalathus linearis (Rooibos tea) as potential phytoremediation agent: a review on tolerance mechanisms for aluminum uptake. Environmental Reviews. 2013; 21(2): 85-92.
CrossRef - Laudicina, V. A., Badalucco, L., Palazzolo, E. Effects of compost input and tillage intensity on soil microbial biomass and activity under Mediterranean conditions. Biology and Fertility of Soils. 2011; 47: 63-70.
CrossRef - Meftah Kadmiri, I., El Mernissi, N., Azaroual, S. E., Mekhzoum, M. E. M., Qaiss, A. E. K., Bouhfid, R. Bioformulation of microbial fertilizer based on clay and alginate encapsulation. Current Microbiology. 2021; 78(1): 86-94.
CrossRef - Bhattacharjee, R., Dey, U. Biofertilizer, a way towards organic agriculture: A review. African Journal of Microbiology Research. 2014; 8(24): 2332-2343.
CrossRef - Calabi-Floody, M., Medina, J., Rumpel, C., Condron, L. M., Hernandez, M., Dumont, M., de La Luz Mora, M. Smart fertilizers as a strategy for sustainable agriculture. Advances in agronomy. 2018; 147: 119-157.
CrossRef - Medina, J., Monreal, C., Barea, J. M., Arriagada, C., Borie, F., Cornejo, P. Crop residue stabilization and application to agricultural and degraded soils: A review. Waste Management. 2015; 42: 41-54.
CrossRef - Khavazi, K., Rejali, F. The use of some low cost materials as carrier for Bradyrhizobium japonicom, Journal of Soil and Water Conservation. 2000; 14(1): 36-45.
- Shilpa, M. E., Brahmaprakash, G. P. Amendment of carrier with organic material for enhancing shelf life of microbial consortium. Journal of pure and applied microbiology, 2016; 10: 2835-2842.
CrossRef - O’connell, P. F. Sustainable agriculture-a valid alternative. Outlook on Agriculture. 1992; 21(1): 5-12.
CrossRef - Jaiswal, S. K., Mohammed, M., Ibny, F. Y. I., Dakora, F. D. Rhizobia as a Source of Plant Growth-Promoting Molecules: Potential Applications and Possible Operational Mechanisms. Frontiers in Sustainable Food Systems. 2020; 4: 619676.
CrossRef - Albareda, M., Rodríguez-Navarro, D. N., Camacho, M., Temprano, F. J. Alternatives to peat as a carrier for rhizobia inoculants: solid and liquid formulations. Soil Biology and Biochemistry, 2008; 40(11): 2771-2779.
CrossRef - Khavazi, K., Rejali, F., Seguin, P., Miransari, M. Effects of carrier, sterilisation method, and incubation on survival of Bradyrhizobium japonicum in soybean (Glycine max L.) inoculants. Enzyme and Microbial Technology, 2007; 41(6-7): 780-784.
CrossRef - Kumar, A., Singh, R., Singh, P. Viability and phosphate solubilization efficiency of phosphate solubilizing bacteria in different carrier materials. Journal of Plant Nutrition, 2022; 45: (12), 1845-1857.
- Gaind, S., Gaur, A. C. Evaluation of fly ash as a carrier for diazotrophs and phosphobacteria. Bioresource technology. 2004; 95(2): 187-190.
CrossRef - Kumar, A., Kushwaha, K. K., Singh, S., Shivay, Y. S., Meena, M. C., Nain, L. Effect of paddy straw burning on soil microbial dynamics in sandy loam soil of Indo-Gangetic plains. Environmental Technology & Innovation. 2019; 16: 100469.
CrossRef - Maheshwari, D. K., Dheeman, S., Agarwal, M. Phytohormone-producing PGPR for sustainable agriculture. Bacterial metabolites in sustainable agroecosystem. 2015; 159-182.
CrossRef - Shilpa, M. E., Brahmaprakash, G. P. Evaluation of selected organic materials as carriers for agriculturally important microorganisms. Mysore Journal of Agricultural Sciences. 2016; 50(2): 266-271.
- Mohammadi, K., Sohrabi, Y. Bacterial biofertilizers for sustainable crop production: a review. ARPN Journal of Agricultural and Biological Science, 2012; 7(5): 307-316.
- Díaz-Rodríguez, A. M., Parra Cota, F. I., Cira Chávez, L. A., García Ortega, L. F., Estrada Alvarado, M. I., Santoyo, G., de Los Santos-Villalobos. S. Microbial Inoculants in Sustainable Agriculture: Advancements, Challenges, and Future Directions. Plants, 2025; 14: (2), 191.
CrossRef - Gaind, S., Gaur, A. C. Shelf life of phosphate-solubilizing inoculants as influenced by type of carrier, high temperature, and low moisture. Canadian journal of microbiology. 1990; 36(12): 846-849.
CrossRef - Viveganandan, G., Jauhri, K. S. Growth and survival of phosphate solubilizing bacteria in calcium alginate. Microbiological Research. 2000; 155: 205–207.
CrossRef - Viveganandan, G., Jauhri, K. S. Efficacy of rock phosphate based soil implant formulation of phosphobacteria in soybean (Glycine max Merrill). Indian Journal of Biotechnology. 2002; 1: 180–187.
- Mahdi, S. S., Hassan, G. I., Samoon, S. A., Rather, H. A., Dar, S. A., Zehra, B. Bio-fertilizers in organic agriculture. Journal of phytology. 2010; 2(10): 42-54.
- Bashan, Y. Inoculants of plant growth-promoting bacteria for use in agriculture. Biotechnology advances. 1998; 16(4): 729-770.
CrossRef - Forestier, S., Alvarado, G., Badjel Badjel, S., Lesueur, D. Effect of Rhizobium inoculation methodologies on nodulation and growth of Leucaena leucocephala. World Journal of Microbiology and Biotechnology. 2001; 17: 359-362.
CrossRef - Bashan, Y., Hernandez, J. P., Leyva, L. A., Bacilio, M. Alginate microbeads as inoculant carrier for plant growth-promoting bacteria. Biology and Fertility of Soils. 2002; 35: 359–368
CrossRef - Gade, R. M., Chaithanya, B. H., Khurade, K. C. A comparative study of different carriers for shelf life of Pseudomonas fluorescens. The Bioscan. 2014; 9(1): 287-290.
- Pratiwi, E., Purwani, J., Yuniarti, E., Husen, E. Effect of polymer additives on viability of Rhizobium japonicum in Molasses-based liquid organic fertilizer. In AIP Conference Proceedings (Vol. 2957, No. 1). AIP Publishing, 2024.
CrossRef - Hindersah, R., Rahmadina, I., Harryanto, R., Suryatmana, P., Arifin, M. Bacillus and Azotobacter counts in solid biofertilizer with different concentration of zeolite and liquid inoculant. In IOP Conference Series: Earth and Environmental Science(Vol. 667, No. 1, p. 012010). IOP Publishing. 2021.
CrossRef - Chenu, C., Stotzky, G. Interactions between microorganisms and soil particles: An overview. In: Huang, P.M., Bollag, J.M. and Senesi, N. (eds) Interactions between Soil Particles and Microorganisms. Wiley and Sons, New York, 2002; pp. 3–40.
- Abd El-Fattah, D. A., Eweda, W. E., Zayed, M. S., Hassanein, M. K. Effect of carrier materials, sterilization method, and storage temperature on survival and biological activities of Azotobacter chroococcumAnnals of Agricultural Sciences, 2013; 58(2): 111-118.
CrossRef - Shravani, K. Evaluation of shelf life and quality of carrier and liquid based biofertilizers. International Journal of Microbiology Research. 2019; ISSN: 0975-5276.
- Novinscak, A., Filion, M. Long term comparison of talc-and peat-based phytobeneficial Pseudomonas fluorescens and Pseudomonas synxantha bioformulations for promoting plant growth. Frontiers in Sustainable Food Systems. 2020; 4: 602911.
CrossRef - Parab, N., Mishra, S., Bhonde, S. R. Mycorrhizal dependency, yield and biochemical parameters of onion in response to fly ash amendment with different biofertilizers. International Journal of Environmental Science. 2013; 2(1): 34-41.
- Gaind, S., Gaur, A. C. Quality assessment of compost prepared from fly ash and crop residue. Bioresource Technology. 2003; 87(1): 125-127.
CrossRef - Juwarkar, A. A., Jambhulkar, H. P. Phytoremediation of coal mine spoil dump through integrated biotechnological approach. Bioresource Technology. 2008; 99(11): 4732-4741.
CrossRef - Pichtel, J. R., Hayes, J. M. Influence of fly ash on soil microbial activity and populations. Journal of Environmental Quality. 1990; 19: 593-597
CrossRef - Selvakumari, G., Baskar, M., Jayanthi, D., Mathan, K. K. Effect of integration of fly ash with fertilizers and organic manures on nutrient availability, yield and nutrient uptake of rice in Alfisols. Journal of the Indian Society of Soil Science. 2000; 48(2): 268-278
- Rama Rao, D., Nanda, S. K., Kareemulla, K. Technological Forecasting and Assessment of Future Fly Ash use in Agriculture in India, DST Project Report, National Academy of Agricultural Research Management, Hyderabad, India. pp 140. 2012.
- Deepti, S., Mishra, S. Exploitation of Fly Ash as a Carrier for Biofertilizers. International Conference on Prospects & Challenges in Biotechnology & Printing and Packaging I Navi Mumbai, 2013.
- Bagchi, S. S., Jadhan, R. T. Pesticide dusting powder formulation using fly ash-A cost effective innovation. Indian Journal of Environmental Protection. 2006; 26(11), 1019.
- Sankari, S. A., Narayanasamy, P. Bio-efficacy of fly ash-based herbal pesticides against pests of rice and vegetables. Current science. 2007; 811-816.
- Dhadse, S., Kumari, P., Bhagia, L. J. Fly ash characterization, utilization and Government initiatives in India. A review. 2008.
- Arivazhagan, K., Ravichandran, M., Dube, S., Mathur, V., Khandakar, R., Yagnanarayana, K., Kamal Pasha, M., Sinha, A., Sarangi, B., Tripathi, V., Gupta, S., Singh, R., Ali, M., Thakur, A., Narayan, R. Effect of coal fly ash on agricultural crops: Showcase project on use of fly ash in agriculture in and around thermal power station areas of National Thermal Power Corporation Ltd., India. World Coal Ash Conference – May 9-12, 2011; Denver, CO, USA.
- Mishra, S., Mishra, D. R., Lee, Z., Tucker, C. S. Quantifying cyanobacterial phycocyanin concentration in turbid productive waters: A quasi-analytical approach. Remote Sensing of Environment. 2013; 133: 141-151.
CrossRef - Ahmad, A., Alam, M. M. Utilization of flyash and Paecilomyces lilacinus for the control of Meloidogyne incognita. International Journal of Nematolology. 1997; 7: 162–164.
- Khan, M. R., Singh. Effects of soil application of fly ash on the fusarial wilt of tomato cultivars. International Journal of Pest Management. 2001; 47 (4): 293–297.
CrossRef - Karpagavalli, S., Ramabadran, R. Effect of lignite fly ash on the growth and dry matter production (DMP) of soil borne pathogens. Souv and abstracts: National Seminor on Bio-Utilization of Fly ash. 1997; 4-5.
- Bhattacharya, S. S., Chattapadhyay, G. N. Transformation of nitrogen during vermicomposting in fly ash. Waste Management & Research, 2004; 22(6): 488-491.
CrossRef - Jeyanthi, A., Balakumar, S., Mahalakshmi, T. Increasing bioavailability of Nutrients in Fly Ash through Vermicomposting. Journal of Bioscience and Technology. 2010; 1(2): 100-113.
- Venkatesh, R. M., Eevera, T. Mass reduction and recovery of nutrients through vermicomposting of fly ash. Applied Ecology and Environmental Research. 2008; 6(1): 77-84.
CrossRef - Yavarzadeh, M. R., Shamsadini, H. Safe environment by using flyash and vermicompost on wheat. International Conference on Transport, Environment and Civil Engineering (ICTECE’2012) August 25- 26, 2012, Kuala Lumpur (Malaysia).
- Polat, M., Lederman, E., Pelly, I. Chemical neutralization of acidic wastes using flyash. Israel. J Chem Technol Biotechnol. 2002; 77(3): 377–81.
CrossRef - Palanichamy, V., Mitra, B., Reddy, N., Katiyar, M., Rajkumari, R. B., Ramalingam, C., Arangantham, A. Utilizing food waste by vermicomposting, extracting vermiwash, castings and increasing relative growth of plants. International Journal of Chemical and Analytical Science. 2011; 2(11): 1241-1246.
CrossRef - Ashwin, R., Bagyaraj, D. J., Kale, R. D. Vermicompost either alone or with amendment can enhance the shelf-life of P and Zn mobilizing fungal inoculants used in sustainable agriculture. Mycosphere, 2016; 7(10): 1526-1532.
CrossRef - Oyege, I., Balaji Bhaskar, M. S. Effects of vermicompost on soil and plant health and promoting sustainable agriculture. Soil Systems, 2023; 7: (4), 101.
CrossRef - Kuppusamy, S., Krishnan, P. S., Kumutha, K., French, J., Carlos, G. E., Toefield, B. Suitability of UK and Indian source Acacia wood based biochar as a best carrier material for the preparation of Azospirillum International Journal of Biotechnology. 2011; 4(6): 582-588.
- Bora, T., Özaktan, H., Göre, E., Aslan, E. M. E. K. Biological control of Fusarium oxysporum sp. melonis by wettable powder formulations of the two strains of Pseudomonas putida. Journal of Phytopathology. 2004; 152(8‐9): 471-475.
CrossRef - Paau, A. S. Formulation of beneficial organisms applied to soil. Formulation of Microbial Biopesticides: Beneficial microorganisms, nematodes and seed treatments. 1998; 235-254.
CrossRef - O’Callaghan, M. Microbial inoculation of seed for improved crop performance: issues and opportunities. Applied microbiology and biotechnology. 2016; 100(13): 5729-5746.
CrossRef - Arangarasan, V., Palaniappan, S. P., Chelliah, S. Inoculation effects of diazotrophs and phosphobacteria on rice. Indian journal of microbiology. 1998; 38(2): 111-112.
- Rajasekar, K., Karmegam, N. Earthworm casts as an alternate carrier material for biofertilizers: Assessment of endurance and viability of Azotobacter chroococcum, Bacillus megaterium and Rhizobium leguminosarum. Scientia Horticulturae. 2010; 124(2): 286–289.
CrossRef - Karunya, S. K., Reetha, D. Survival of saline tolerant PGPR in different carriers and liquid formulations. International Journal of Advanced Research in Biological Sciences. 2014; 1(2): 179-183.
- Singh, S., Prasad, S. M. Growth, photosynthesis and oxidative responses of Solanum melongena seedlings to cadmium stress: mechanism of toxicity amelioration by kinetin. Scientia Horticulturae. 2014; 176: 1-10.
CrossRef - dos Reis, G. A., Martínez-Burgos, W. J., Pozzan, R., Pastrana Puche, Y., Ocán-Torres, D., de Queiroz Fonseca Mota, P., … & Soccol, C. R. Comprehensive Review of Microbial Inoculants: Agricultural Applications, Technology Trends in Patents, and Regulatory Frameworks. Sustainability, 2024; 16: (19), 8720.
CrossRef - Cigdem, K., Merih, K. Effect of formulation on the viability of biocontrol agent, Trıchoderma harzıanum. African Journal of Biotechnology. 2005; 4(5): 483–486.
- Walker, B., Holling, C. S., Carpenter, S. R., Kinzig, A. Resilience, adaptability and transformability in social–ecological systems. Ecology and society. 2004; 9(2).
CrossRef
Abbreviations
| PGPR | Plant-growth-promoting rhizobacteria |
| EC | Electrical conductivity |
| FYM | Farmyard manure |
| SMS | Spent mushroom substrate |
| CFU | Colony forming units |


